No Rub, no Cone: The Keratoconus Conjecture.
No rub No cone
by Damien Gatinel, MD, PhD, from the Rothschild Foundation, Paris, France.
The word keratoconus originates from the Greek word “kéras” meaning cornea and the Latin word “cōnus” meaning cone. This denomination is purely descriptive however and does not suggest any mechanism to account for the corneal deformation which remains the hallmark of the disease. The progressive structural deformation of the corneal shape leads to refractive instability and reduction in the optical quality of the keratoconic eye. The exact molecular or tissue abnormalities in keratoconus are still unknown.
Eye rubbing is a common activity occurs at different times of the day: most regularly upon waking, before sleep, during extended computer work, and throughout the day in response to ocular itch and irritation, fatigue or emotional stress (1,2). It is usually benign but when performed too vigorously, too frequently, or over extended periods, rubbing becomes pathological and can be harmful to the cornea. Prolonged and repetitive forceful knuckle rubbing is often seen in keratoconus.
In this paper, I wish to defend a new theory in which abnormal eye rubbing is not just a risk factor, but is the main and necessary causative factor of keratoconus (3). In this postulate, therefore, the absence of rubbing would prevent keratoconus from occurring and this conjecture may be condensed as: “no rub, no cone”.
In mathematics, a conjecture is a proposition which is consistent with known data. It has neither been verified nor shown to be false. Whilst formal proof that eye rubbing is the indispensable causative factor for keratoconus may be difficult, « no rub, no cone » represents a valid conjecture for elucidating the mystery of the pathophysiology of keratoconus, therefore offering potential benefits to the management of what is considered the most frequent corneal “dystrophy”.
Introduction
The blunt statement “that eye rubbing is a chronic mechanical injury necessary to trigger and accelerate the corneal deformation observed in keratoconic eyes” defies the widely adopted theory that keratoconus is primarily a corneal dystrophy. However, it is entirely possible that keratoconus may truly be a primary mechanical disease and in fact, I would submit that this mechanical theory provides a better explanatory framework for what is currently known about keratoconus (KC). This theory does not contradict previous clinical or experimental findings of KC, all of which can be interpreted in light of this proposed mechanism. Rather, it is a synthetic approach that incorporates the results of previous genetic and biochemical approaches for understanding the pathophysiology of keratoconus. In fact, the mechanical disease proposition would appear more compatible with explaining the variability of keratoconus expression between patients, between eyes and the predominance of sporadic cases.
Beyond academic debate, the “no rub, no cone” conjecture suggests that there is a possibility of stopping the progression of existing keratoconus by the cessation of eye rubbing. Even more importantly, if the mechanical injury caused by chronic eye rubbing really is a necessary ingredient in the recipe of the keratoconus disease, surely its suppression would make it possible to even eradicate it. As such, it should raise significant interest among the medical and ophthalmic community.
A conjecture is considered proven only when it has been shown that it is logically impossible for it to be false. Whilst mathematicians would seek robust demonstrative proof to make it a theorem, in contrast, new theories in medicine can be supported by conjectural models relying on tangible evidence. In what follows, I will show why the “no rub, no cone” conjecture may be proven correct by existing logical data and evidence. The line of arguments developed here will hopefully receive enough consideration to be accepted as a viable theory and, if proven true, lead to new guidelines for managing and preventing keratoconus.
Keratoconus is not an ectasia but a permanent corneal warpage
The corneal deformation in keratoconus is often referred to as an “ectasia”, but this term is not specific to keratoconus and is also employed to describe the most dreaded complication of LASIK surgery.
In medicine, an ectasia is defined by a permanent widening, distension or ballooning of any tubular organ or part, or more specifically, “a dilation, expansion, or distension” (4), all of which invokes the notion of an increase in surface area by a process of stretching. Annulo-aortic ectasia, for example, refers to a proximal dilatation of the aortic root at the level of the aortic annulus. The diagnosis is based on the increase in aortic wall diameter. It occurs with conditions such as Marfan disease, Ehlers-Danlos syndrome and other familial forms of connective tissue diseases.
In the ophthalmic context, the use of the term “corneal ectasia” is a misnomer as it is not a truly accurate description of the morphology of the keratoconic cornea. An ectatic bulge would necessarily incur an increase in surface area of the anterior corneal surface. However, in 2000, Smolek and Klyce demonstrated that the surface area of the cornea does not, in fact, increase when keratoconus develops (5). Except for the single case of keratoglobus, corneal surface area tended to be conserved near a value of 120 mm2 for all groups in their study, including corneas with keratoconus. The authors reported that the surface area is remarkably insensitive to curvature change near the vertex. Thus, they concluded that keratoconus is not a true ectasia (unlike keratoglobus where the corneal surface expands), but rather a specialized type of warpage, at least in mild to moderate forms of the disease.
From a mathematical perspective, Gauss’ Theorema Egregium (Latin for « Remarkable Theorem ») is a foundational proof in differential geometry demonstrated by Carl Friedrich Gauss concerning the curvature of surfaces. His theorem states that the Gaussian curvature of a surface does not change if one bends the surface without stretching it (6). Hence, the flattening seen in the periphery of keratoconic corneas (increased prolateness) corresponds to a necessary coupling effect compensating for the increase in curvature in the cone region. These observations bluntly contradict the concept of keratoconus being an ectasia in the strict medical sense and indicate that the deformation of keratoconic corneas may be better described as an extreme but isometric form of distortion or warpage, which redistributes the corneal curvature without local protrusion.
The common perception that keratoconic corneas are “ectatic” may be partly due to the subjective interpretation of curvature maps obtained from specular corneal topographs (Figure 1).
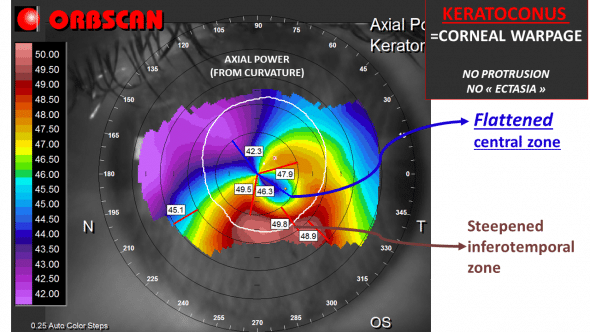
Figure 1: Axial power map of a keratoconic cornea (left eye). The inferotemporal red zone can be interpreted by the inexperienced physician as an area of local protrusion. However, this map does not correspond to a plot of the shape of the cornea. Rather, it corresponds to the representation of the local axial curvature of the anterior corneal surface. Curvature is inversely proportional to the local axial radius of curvature (the shorter the radius, the steeper the surface). This inferior steepening does not mean that the corneal surface is bulging inferiorly. Rather, the deformation of the cornea incurs a predominant central flattening along an oblique direction, which is the result of the curvature coupling effect compensating for the inferior steepening. The mean gaussian curvature within the central 8 mm diameter zone is balanced to 40D (a value similar to what would be encountered in normal corneas), which is consistent with the Gauss’ Theorema Egregium.
Using conventional topography scales, the increase in axial curvatures plotted in maps of keratoconic corneas results in islands of warmer colors surrounded by annuli of cooler hues. Our subjective visual perception tends to isolate any area colored in red from the surrounding zones of bluer colors, and this distinction may lead to a false impression of a local protrusion. The colors and scales used in corneal topography, however, are arbitrarily chosen and when their parameters are varied, may lead to variations in the plot of the same data. Hence it would make no sense to delineate a “cone” by simply following the contours of an area of reddish colors. Moreover one should not try to extrapolate the three-dimensional shape of the cornea from axial curvature plots and consider any “red zone” as equivalent to a local bulging.
The deformation of the keratoconic cornea is global and not confined to the central area, despite what the color renditions of axial curvature maps may incorrectly suggest. The inspection of the corresponding elevation map of such surfaces, which is a truer representation of the actual shape of the considered corneas, allows one to rectify this impression and reveals a more progressive gradient of colors (Figure 2).
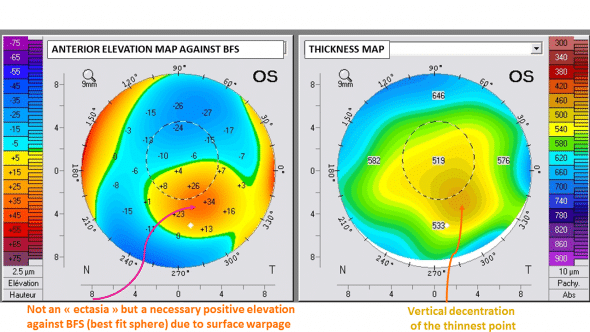
Figure 2: Left : the anterior elevation map provides a better representation of the spatial properties of this anterior surface. The inferiorly decentered island of red colors corresponds to positive elevation against the sphere declivity. It is a consequence of global corneal deformation, and should not be interpreted as a local protrusion.
Right: The thickness (pachymetry) map reveals the temporal inferior thinning of the cornea, which is accompanied by a vertical displacement of the thinnest point.
Elevation plots against a best-fit sphere generally outline an “island pattern” which reflects the increased prolateness of the keratoconic surface (decrease in the curvature from the corneal apex to the periphery) (7). In keratoconus, the corneal deformation is characterized by a warpage combining a central steepening with a concomitant compensatory peripheral flattening. The complete absence of any associated dilatation or expansion makes the term “ectasia” inappropriate in describing accurately the morphological changes at play in the keratoconic cornea (Figure 3 and 4).
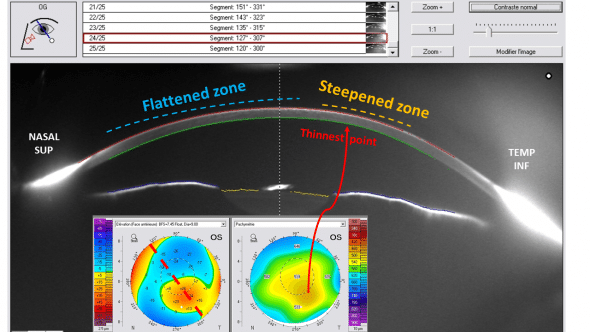
Figure 3: This oblique cross section of the Scheimpflug image provides a direct visualization of the cornea deformation and notably does not suggest any local« protrusion » or « ectasia ». Rather, the corneal deformation results in a different repartition of the curvature along this particular corneal meridian: the slight infero-temporal flexure of the cornea is adjacent to the thinnest part of the corneal, and is accompanied by a concomitant supero-nasal flattening.
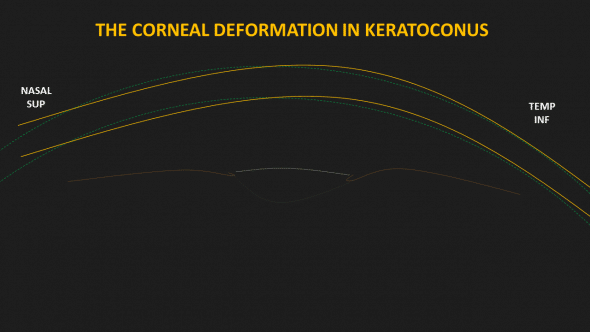
Figure 4 : This is a schematic representation of the global deformation of a keratoconic cornea. Many synergistic mechanisms brought about by pathological rubbing induces permanent warpage. This deformation is caused by a paracentral flexure of the corneal wall accompanied by a central thinning which is mainly caused by a displacement of the ground surface of the cornea during the initial phase of the disease.
In keratoconus, the posterior corneal curvature is affected in addition to the anterior corneal surface. Moreover, early morphological changes in eyes with keratoconus may develop on the posterior surface. In a theoretical study, we established that the representation in elevation of theoretical corneal models against a best-fit sphere (BFS) may explain the differences in the elevation patterns of the anterior and posterior corneal surfaces (7). Given the geometry of the posterior corneal surface, which is naturally steeper and more aspheric than the anterior surface, any concomitant apical steepening and peripheral flattening of both corneal surfaces would result in a greater increase in the apical distance to the posterior surface’s best fit sphere (BFS), and therefore a warmer central color in the elevation plot (Figure 5).
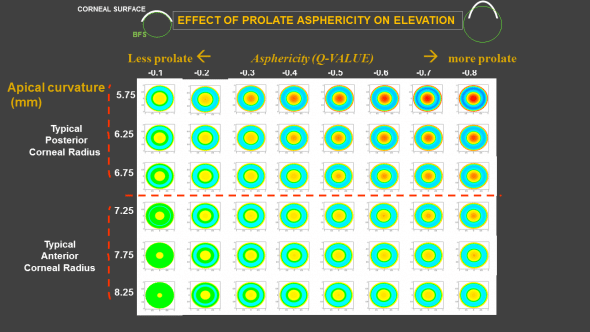
Figure 5 : This figure is showing various theoretical corneal surfaces, which were modeled as aspheric surfaces of different elliptical profiles, all plotted in elevation against their best fit sphere (BFS). The evolution of keratoconus corresponds to an increase in a corneal deformation which is characterized by an apical steepening, and an increase in negative asphericity (increased prolateness). For the same asphericity change, the steeper surface will have more central elevation against the BFS (and warmer colors on the elevation map). This is due to mathematical and scaling reasons. The keratoconic deformation does not start posteriorly in the cornea, but its concomitant effects on the anterior and posterior corneal surfaces elevation plots are not linear and more perceptible on the posterior map.
These theoretical predictions echo the clinical impression that early manifestations of keratoconus are often characterized by an increase in the apical posterior elevation. Our results show that during the evolution of keratoconus, the posterior surface of the cornea deforms concomitantly with the anterior surface of the cornea, but that the effects of these changes on elevation maps are more visible in the posterior surface of the cornea.
What then causes the corneal warpage of the keratoconic cornea?
Whilst both environmental and genetic factors are thought to contribute to the development of the disease, the physiopathology of keratoconus has not been elucidated until now. An exploration of the supportive and intertwining sub-theories follows.
The case revisited – Are these risk factors correlation or causative?
Eye rubbing has been presented as a risk factor (by correlation) for keratoconus, amongst others including ocular allergy, atopy, and Down syndrome (8-10) Keratoconus appears more prevalent in certain ethnicities (11,12). It has also been reported that diabetes could be a protective factor against keratoconus (9).
Much of scientific evidence, however, is based upon a correlation of variables. It is, therefore, necessary to point out that correlation does not necessarily mean causation. When two variables are found to be correlated, it is tempting to assume that this demonstrates that one variable causes the other. A similar fallacy would state that an event that followed another one was necessarily a consequence of the first event.
For instance, the well-established correlations between keratoconus and atopy or Down-syndrome may not mean that atopy or Down-syndrome directly causes keratoconus. A spurious relationship may explain both correlations, because in reality, eye rubbing may be the confounding variable. In the context of atopy or Down syndrome, chronic eye rubbing may be the reason for increased risk for keratoconus. In such a context, the correlation between chronic eye rubbing and keratoconus corresponds to a direct causative link, not just a statistical correlation.
The case for rubbing and unilateral keratoconus
Unilateral keratoconus has been found to develop only in the eye that is subjected to frequent and abnormal (severe) episodes of rubbing trauma (13-15). These cases and others (16,17) suggest the possibility that KC can develop in a normal eye in response to a single aetiological mechanism such as eye-rubbing related trauma.
The case for rubbing and contact lenses
Patients with keratoconus reported significantly more rubbing before and after wearing their contact lens as well as longer rubbing episodes, compared to non-keratoconus patients wearing contact lenses (18). During the wear of contact lenses, rubbing may induce lacrimal gland expression of more tears to relieve dry eye symptoms. Irritation, dryness, or itching, after lens removal at the conclusion of the day may trigger eye rubbing.
The case from a baropathic perspective
That repeated and vigorous eye rubbing (primarily a mechanical force) may, in fact, be eventually causing the direct change in corneal shape (a permanent deformation) has the merit of being a straightforward concept (19). Using an ophthalmodynamometer to applanate the inferior sclera through the lower lid, researchers have compared the effect of an elevation of intraocular pressure to an average level of approximately double the baseline (20). An average response of 1.84 D of steepening in keratoconic subjects was observed when comparing corneal topography examinations before and after the pressure elevation. No significant topographic changes were observed for eyes with normal corneas. These findings suggest that keratoconic corneas are weakened and unable to support the IOP, the effect of which causes the cornea to bend centrally and flatten peripherally. Hence, elevations in IOP have the potential to contribute to the progression of these conditions and acute events such as hydrops and perforation may be dependent on acute IOP elevations.
For the first time, we have provided direct imaging of the impact of ocular rubbing on the eyeball and adjacent structures:
Moreover, the IOP spikes occurring during regular rubbing episodes may not only aggravate but initiate the weakening of the corneal tissues. The pressure exerted on the corneal wall during vigorous rubbing by patients with keratoconus has been estimated to be 10 times more pronounced than in non keratoconic patients. It can reach 4 Kg per cm2 (21) which is considerable (this, in fact, represents the internal pressure of a car tire!).
Muller et al. showed that the tightly interwoven collagen fiber network in the anterior stroma is mainly responsible for the structural integrity of the cornea (22). The interweaving appears maximal at the anterior surface and significantly reduces toward the posterior stroma (23). Repeated shearing and torsional forces may induce the loosening of this network and reduce the shear modulus which measures the resistance of the corneal tissue to shearing strains. In addition, there may be additive complex effects related to chronic eye rubbing, as the corneal trauma may arise from a combination of possible mechanical effects: high hydrostatic tissue pressure, thixotropically reduced ground substance viscosity, temporary displacement of ground substance from the corneal apex, buckling and flexure of fibrils associated with waves of corneal indentation, and biomechanically-coupled curvature transfer to the cone apex.
A mixture of these factors, like the displacement of ground substance from the corneal apex, may explain the central thinning which is not involved in typical warpage but is instead a constant finding in keratoconic corneas.
When a mechanical stress is removed from a viscoelastic material, there is an instantaneous recovery of the elastic deformation, followed by a slow recovery of the viscous creep. One must realize that corneal tissue itself is viscoelastic: and all the mechanisms evoked previously, all of which have been admirably detailed by the work of McMonnies (24), may, therefore, alter the viscoelastic properties of the cornea. Abnormal rubbing with longer duration and/or greater force and/or greater frequency may involve permanent central buckling of the corneal tissue with compensatory peripheral flattening. Rubbing related temperature spikes may also upregulate further collagenase activity and alter the ground substance viscosity.
These combined effects facilitate the bending response of the cornea to rubbing mediated IOP distending stress via a reduction in its shear modulus (25) and the changes in extracellular matrix (24).
The case from a cellular physiology perspective
Repeated eye rubbing over weeks or months with great force may cause significant corneal tissue responses not limited to direct injury to the collagen fiber arrangement and extracellular matrix but also impact the cellular composition of the corneal tissue. Cells of living organisms have a repertoire of strategies for dealing with mechanical stimuli. Several are relevant to the eye and to the way ocular cells respond to their physical environment in health and disease. This mechanosensitivity is relevant to the eye with respect to axial myopia and the pressure related disease of glaucoma. When subjected to mechanical forces such as compression, extension, torsion, and shearing, human cells can be damaged and become necrotic or apoptotic (26). In addition, increased hydrostatic pressure caused by eye rubbing may increase the apoptotic rates for keratocytes. The subsequently reduced maintenance of collagen and ground substance may reduce further the biomechanical resistance of the cornea.
The case from a cellular inflammation perspective
Eye rubbing for 60 seconds has been demonstrated to increase the level of tear film MMP-13, IL-6 and TNF-α in normal study subjects (27). The increase in protease release, protease activity and inflammatory mediator formation after eye rubbing may be exacerbated even further during the persistent and forceful eye rubbing seen in people with keratoconus. Sustained elevated levels of inflammatory mediators in the tears due to repeated rubbing episodes may be significant in keratoconus development and progression (28).
It has been shown both in animal and human models that there exists a rubbing-related mechanical epithelial trauma which triggers the release of inflammatory mediators and a wound healing response in keratocytes. Slight rubbing for 10 seconds using one finger and in a smooth circular movement repeated 30 times over a 30-minute period was shown to significantly reduce the keratocyte density in human corneas, and also leads to a greater concentration of inflammatory mediators in the tears (29). Rubbing-related epithelial thinning may include cell flattening, as well as displacement from the rubbed area of some cells, extracellular fluid, mucin, and cytoplasm from any burst cells. After an experimental fifteen seconds of rubbing in a circular pattern with use of light to moderate force and the finger pad of an index finger, the epithelial thickness of normal human corneas was found to be reduced by 18.4%, both centrally and mid-peripherally (30). Recovery to baseline thickness took 15 to 30 minutes centrally and 30 to 45 minutes mid-peripherally.
Patients with atopic dermatitis and mild blepharoconjunctivitis without any keratopathy may have an impaired ocular surface epithelial barrier (31). This may facilitate cellular infiltration from tears or other corneal inflammatory processes (32) Since eye rubbing can induce inflammatory events, (27,33), any disease or wound healing process with an inflammatory component has the potential to be exacerbated by rubbing trauma.
The case from a structural microscopy perspective
Cumulative tissue changes caused by repetitious rubbing episodes may also help link chronic rubbing-related trauma to cone formation. Cone deformation commences most often in the paracentral inferonasal cornea, which corresponds to the location where most senile epithelial cells may be more vulnerable to mechanical forces (34). The fine reticular scars of Bowman’s membrane tears preceded by visible dehiscences is a classic characteristic feature of KC and are at this level (35). These dehiscences followed by scarring may represent evidence of rubbing related ruptures of Bowman’s layer.
These mechanisms eloquently suggest that even in the absence of any primary ocular disease, keratoconus can be self-induced as a manifestation of compulsive rubbing behavior. This makes pathological eye rubbing the foundation on which explain the pathophysiology of keratoconus.
Can the cornea deform without repeated focal trauma?
The obligation of repeated local corneal mechanical trauma inducing a permanent corneal deformation is the core of the “no rub, no cone” conjecture. In order to refute this, it would suffice to demonstrate that corneal deformation could occur without any external force, through the reduction of corneal resistance secondary to a molecular alteration of its collagen fibrils.
There is a common assumption that alterations in corneal collagen make the cornea thin and less resistant to the gradient of pressure between the anterior chamber and the atmosphere. An unknown stromal collagen degeneration may indeed make the cornea less resistant, but the mechanisms by which the corneal deformation could occur progressively remains unclear. Although there is no identified genetic mutation nor clear biochemical cascade leading to such change in the corneal shape, would such corneal tissue “softening” due to a hypothetically altered molecular constitution lead to the evolution of keratoconus, without the exertion against the cornea of any other force than that mediated by the difference between intraocular and atmospheric pressure?
The case study with Marfan’s syndrome
Marfan syndrome, a disorder with an identifiable genetic mutation of the fibrillin-1 molecule, represents a perfect counter-example to explain the inconsistency of such theories on the pathogenesis of keratoconus (36). Despite the reduction of collagen strength present in the corneal stroma, no keratoconic pattern is found in the eyes of Marfan syndrome patients. Although these corneas are thinner, they tend to be flatter rather than steeper (37).
There is no paradox here: because the force resulting from IOP is evenly distributed against the posterior surface of the cornea and the inner surface of the sclera, in the absence of a focal stress, a “stromally”-softer eyeball will undergo progressive generalized distension of its shell. This would naturally cause the corneal curvature to decrease, with concomitant progressive thinning. This description better corresponds to what should be truly called an ectatic cornea, since, as for the ectatic aorta, the progressive homogenous stretching and expansion of such a structure leads to an increase in its respective radius of curvature (meaning that the curvature logically decreases).
In Marfan syndrome, morphologic abnormalities resulting from fibrillin-1 mutations can produce the displacement of the crystalline lens but fails to produce any corneal changes other than flattening and thinning. This may sound counter-intuitive to most ophthalmologists who would assume that the thinning and excessive weakening of the corneal stroma « must” result in a central steepening. However, this strongly suggests that, without an additional factor, altered biomechanics from collagen abnormalities alone are insufficient to account for the steepening and weakening seen in keratoconus. Hence, the “no rub, no cone” conjecture is not contradicted. On the other hand, it may be logically postulated that patients with Marfan syndrome or any other related connective tissue disease would be prone to develop keratoconus if they rub their eyes too frequently, given the technical structural weakness of the corneal wall.
In the case of keratoconus, recent studies have demonstrated that the alteration of the corneal curvature and biomechanics are not evenly distributed throughout the corneal wall but are instead concentrated within the corneal apical area. Working with donor corneas, researchers demonstrated using Brillouin measurements that biomechanical alterations in the keratoconic corneas were primarily concentrated within the area of the cone. Outside the cone, the biomechanical properties evaluated with Brillouin measurements were comparable with that in normal corneas (38)! This focal aspect does not favor the hypothesis of unknown collagen dystrophy, which would affect the stroma globally. Conversely, chronic eye rubbing, (which is exerted in the vicinity of the corneal apex where the cascade of tissular changes described previously would predominate) is compatible the focal aspect of damage of the corneal stroma.
Re-examining the “genetics” of keratoconus
Some familial cases and genetic research are still in favor of a genetic dimension to keratoconus but as yet, no genetic mutations have been identified for any of the identified keratoconus chromosome loci. So far, the pattern of inheritance is estimated to be less than 20%. Discordance for keratoconus in two pairs of monozygotic twins has been reported (39) which supports a pathogenetic role for environmental influences such as eye rubbing. Concordance for keratoconus has also been reported between monozygotic twins (40). This concordance may also be due to being in similar environments and confounding variables such as atopy leading to ocular itch and eye rubbing. Neither have pathogenic mutations nor chromosomal deletions/duplications been found to provide a complete explanation for isolated (sporadic) cases of keratoconic patients (41). In essence, it is difficult to explain with genetics the inter-eye variability in the stage of keratoconus and the different ages of onset. One must then ask – could eye rubbing be the necessary non-genetic variant or epigenetic factor to explain the disease etiology?
As alluded to above, genetic components could actually be related to the predisposition of conditions that lead to increased eye rubbing and to variations in corneal thickness and resistance such as Down syndrome, Tourette syndrome, atopy, sleep apnea and sleep disorders, and pregnancy. A cornea that is genetically weaker or constitutionally thinner may be more vulnerable to any rubbing trauma as previously discussed. The central corneal thickness is one of the most highly heritable human traits. Ethnic-related differences in central corneal thickness may partly account for the increased prevalence of keratoconus in some ethnicities (42). Amongst the identified sixteen new loci associated with central corneal thickness, two were found to confer relatively large risk for keratoconus (43). For the same rubbing pattern, thinner corneas may be at greater risk for keratoconus.
Despite intensive research, no specific gene has been discovered and it is highly probable that none will ever be. Rather, efforts conducted in studying molecular genetics to identify people with native thinner or weaker corneas and prone to immune system disorders leading to allergy or atopy should be encouraged.
The role of acquired risk factors for eye rubbing and keratoconus
Over the last few decades, acquired risk factors for eye rubbing and corneal weakening have come to the fore.
Allergy and atopy may be the most commonly addressed provocative factors for abnormal eye rubbing (44). Many epidemiological surveys, amongst which repeated cross-sectional surveys have most validity, have demonstrated a twofold increase in the prevalence of allergic and asthma during the past two decades (45,46). Among allergens, house-dust mites have been advocated to be responsible for the increasing trend in the prevalence of allergic diseases (47). In a sample of 82 consecutive cases of vernal keratoconjunctivitis, which is characterized by intense itching, computer-assisted videokeratographic maps indicated that 27% of eyes showed at least early signs of keratoconus (48).
While air pollutants from industrial sources have been drastically reduced in the last decades, air pollutants from vehicle exhausts (nitrogen dioxide, diesel exhaust particulates, ozone) show some increase in concentration in some urban areas. Experiments have shown a synergy between air pollutants and allergens (49). The risk of developing sensitization to an allergen is boosted when the exposure to air pollutants has been performed prior to the exposure to the allergen.
The tremendous increase in computer usage has led to the development of computer vision syndrome in many individuals. Episodes of rubbing during extended computer use are frequently admitted by patients once properly informed about it. Ocular fatigue and eyestrain are the main components of computer vision syndrome and may elicit pathological eye rubbing (50). Associated evaporative dry eye from reduced blink frequency can also elicit eye rubbing and account in part for the recent increase in the prevalence of keratoconus in the young population.
Contact lens wearers often disclose that they periodically rub their eyes after contact lens removal. Eye rubbing after contact lens removal (‘removal-relief’ rubbing) was found to be significantly more prevalent among contact lens-wearing keratoconic patients compared to contact lens-wearing non-keratoconic patients (18).
Post LASIK ectasia could be the consequence of the stromal flap cut and laser photoablation on corneas already weakened by chronic eye rubbing. Dry eye exacerbation secondary to LASIK may induce rubbing behavior which dramatically increases the risk of corneal ectasia. It is therefore logical that eye rubbing avoidance may reduce the incidence of post-refractive corneal ectasia.
The recent increase in prevalence of these acquired risk factors for eye rubbing may account for the increase in the prevalence and annual incidence of keratoconus (51).
Forme fruste keratoconus – Forme fruste rubbing – the missing link?
In the surgical field, the identification of subclinical keratoconus is a primary concern when screening patients for refractive surgery because performing laser-assisted in situ keratomileusis (LASIK) on undiagnosed keratoconus has been identified as the leading cause of ectasia after refractive surgery. Several terms have also been used to describe this condition, including subclinical keratoconus, keratoconus suspect, and forme fruste keratoconus. Beyond any semantic debate, what these entities have in common in terms of topography is that they share the faded features of corneas which we diagnose as having keratoconus. In the light of the conjecture, they correspond to the missing links between corneas that are strictly normal (never or rarely rubbed), and corneas very distorted (by repeated and pathological rubbing), which correspond to established keratoconus.
In our practice, there is indeed remarkable clinical parallelism between these slightly deformed corneas which cause diagnostic problems where the practice of eye rubbing is not very intense or less frequent. For example, some women remove their eye make-up every night and rub their eyes daily vigorously on this occasion. This habit may cause moderate corneal deformations which cause the apparition of topographic patterns such as an isolated inferior steepening. Due to the risk of smearing eye cosmetics, these women tend to avoid rubbing their eyes during the day, which prevents the corneal deformation from becoming more severe more rapidly. In contrast, men are more likely to develop frank keratoconus.
In short, forme fruste keratoconus may simply correspond to “forme fruste rubbing” (Figure 6). The large variations in rubbing intensity, frequency and duration among patients and their effects on corneas of variable native resistance may be reflected in the broad topographic spectrum of warpage and thinning; from minor topographic abnormalities to advanced keratoconus, encompassing all shades of subclinical keratoconus in between.
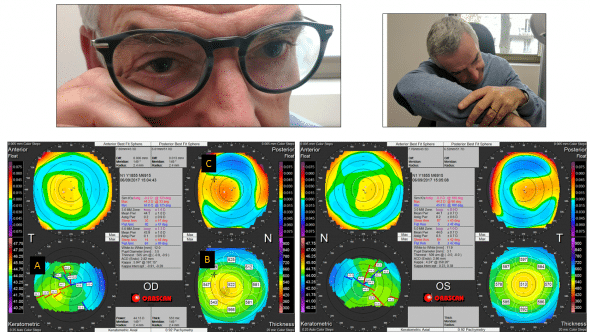
Figure 6: This 55 yo patient has been referred for refractive surgery evaluation (correction of hyperopia). Minor topographic abnormalities are seen on the right eye topography: a slight inferotemporal steepening (A), and a paracentral inferior thinning (B). The posterior surface prolateness is increased (C). The left eye topography is unremarkable. The right eye topographic features are commonly found in forme fruste keratoconus. When asked about possible chronic rubbing habits, the patient admits that he wipes his right eye often during the day, because this eye feels irritated and watery. When questioned about his sleeping habits, the patient declares that he constantly sleeps on the stomach, with the head rotated on the left, causing the right orbit to be compressed on the right forearm.
In our experience, there is a striking correlation between the side on which patients sleep, and the side where the most pronounced topographic abnormalities are evident. Compression, heating and local contamination may all contribute to ocular irritation at night, leading to itching sensations during the day. The itch in turn triggers repeated eye rubbing leading to various levels of deformation depending on native corneal properties and the duration, frequency, type, and intensity of eye rubbing.
The displacement of the thinnest corneal area may occur early in the pathologic process. In a previous study (52), we showed that the thinnest corneal width position and the vertical decentration of the thinnest corneal thickness was statistically different between normal corneas and forme fruste keratoconic corneas (the forme fruste keratoconic group was composed of corneas appearing normal based on a specular artificial intelligence system, while the fellow eye had frank keratoconus). However, we also found in another study that there was no inter-eye difference regarding either the thinnest corneal thickness position or the vertical decentration of the thinnest corneal thickness between keratoconic eyes of various stages (53).
These findings support the hypothesis that thinnest corneal thickness decentration occurs early in the keratoconic pathologic process. The downward displacement of the thinnest point may be caused by two synergistic mechanisms. The first may be the consequence of the upward and outward movement of the eye when the eyes close (this palpebral oculogyric reflex corresponds to Bell’s phenomenon which is thought to be a defensive phenomenon) (54). This may preferentially expose the inferior part of the cornea to the rubbing forces, which are exerted on closed eyes. The displacement of the ground substance may be maximal near but inferior to the corneal apex. The second mechanism may be related to the downward direction of the gravity force: one may also hypothesize that this force may induce some asymmetry in the flexure of the weakened corneal wall, which would also incur a more inferiorly pronounced steepening. Even without any spatial redistribution of the corneal thickness, this would cause an apparent slight inferior displacement of the thinnest point from its initial location due to the vertical “sag” of the corneal dome.
Eye rubbing is the root cause of keratoconus
Bringing all of these elements together, a new model to explain keratoconus formation emerges. Based on these findings, eye rubbing is detrimental to corneal biomechanical stability through two main pathways, which are synergetic as both reduce the resistance of the cornea (Figure 7). The first pathway outlines the mechanical impact of rubbing on tissue structures such as collagen fibrils. Rubbing-related buckling and flexure of these fibrils may facilitate cone formation, associated with fibrillar slippage in the cone region. The second pathway corresponds to the impact of eye rubbing on cellular structures, which may undergo changes and apoptosis, further compromising corneal structural properties.
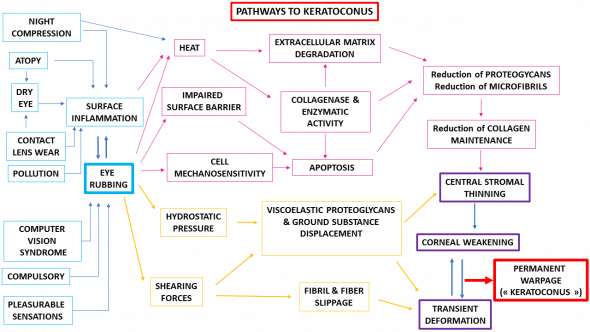
Figure 7 : Eye rubbing is a central and obligatory habit that induces the permanent warpage of the cornea referred to as « keratoconus ». Without eye rubbing, the absence of increased hydrostatic pressure and shearing forces will not result in significant corneal deformation (no keratoconus). Interestingly, inflammatory processes in the cornea usually result in localized topographic flattening. The imbalance between the reduced corneal resistance and abnormal rubbing eventually causes permanent warpage of the cornea.
Does a keratoconic eye always have a rubbing history?
Finding a counterexample to the conjecture would suffice to invalidate it. Conversely, for this theory to be valid, it should be demonstrated that every patient presenting with keratoconus has a history of chronic eye rubbing. Although such a systematic approach may be difficult to establish, our experience at the Rothschild foundation is amazingly conclusive.
While most patients disclose a rubbing history (often associated with a chronic ocular allergy history) on their first visit, some patients are not conscious of their rubbing habit and when first interviewed will reply in the negative to the question, “Are you rubbing your eyes frequently?” (Figure 8). If asked too bluntly, this question may sometime destabilize the patient-doctor relationship and elicit some falsely negative answers. Some patient may mistakenly believe that their physician is trying to blame the visual problems on the patient, and be reluctant to disclose their rubbing habit. The patient’s denial of a history of rubbing could also be based on guilty feelings about potentially inflicting injury to oneself. Undisclosed rubbing may occur during sleep or without awareness. The presence of close relatives accompanying the patient offers the possibility to confirm or confront the patient’s declaration. Frequently, parents often vehemently declare that they have witnessed their child rubbing their eyes repeatedly in the past.
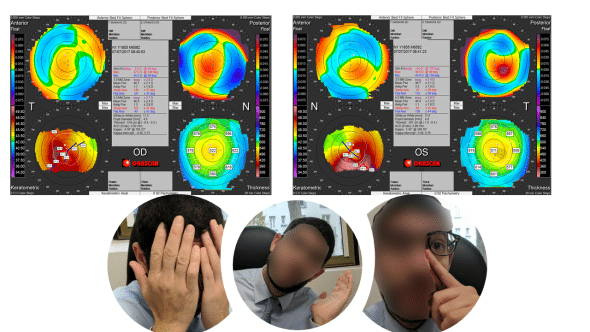
Figure 8: Top: corneal topographies of the right (OD) and left (OS) of a 40 yo myopic patient referred for refractive surgery preoperative evaluation. These maps, obtained with the Orbscan topograph, reveal the presence of an unknown bilateral keratoconus pattern, slightly more pronounced on the left eye. During this first visit, the patient did not acknowledge any particular eye rubbing habit. We explained to this patient that eye rubbing could occur without self awareness, and asked him to pay attention to possible unknown rubbing episode in the coming days.
Bottom: On his second visit, one month later, the patient declared that he realized that he rubbed his eyes many times a day, using his hands palms, or the index pulp and knuckles. He spontaneously rubbed his left eye to demonstrate it. He also declared that he was always sleeping on the left side, with his head buried in the pillow. He attributed this rubbing habit to fatigue and overwork on the computer. His sleeping habit may also explain the preferential rubbing of the left eye.
In a pioneer study published in 1976, 40 out of 55 (72%) patients with keratoconus closely questioned about eye-rubbing gave a positive response (55). This study reports that some of these denied any personal knowledge of eye rubbing but remembered mothers or others reprimanding them for it. The paper also cites the example of a patient with eczema who denied eye-rubbing or skin-scratching, yet there were obvious scratch marks on facial eczematous lesions. A study of the prevalence of eye rubbing found that it was reported in 80% of patients with keratoconus, and in 58% of control subjects, with the difference being significant (P<0.001) (56). Based on these numbers, is not unreasonable to postulate that about 20% to 28% of patients with keratoconus would not be aware, or would not voluntarily disclose a chronic rubbing habit at the first glance. This denial would account for the several studies indicating “no history of abnormal rubbing” for some patients with keratoconus (57), leading to the possibly erroneous conclusion that the causal rubbing hypothesis may be appropriate for only some forms of keratoconus.
Searching for eye rubbing
In my practice, every patient evaluated for keratoconus receives information intended to increase their awareness of possible unconscious rubbing episodes, which typically occur during awakening times, under the shower, during extensive computer work (or video games), after contact lens removal, and before falling asleep. They are, of course, advised to refrain from any vigorous rubbing when this is understood. All patients are systematically reviewed one month after the first visit for a control examination. During this second interview, the propensity to chronic eye rubbing is re-evaluated. In our experience, all the patients who at first denied eye rubbing acknowledged that they were not aware that they were eye rubbers. Amongst those who disclosed this habit, many declared that they were now actively minimizing it. While atopy and ocular itch are only being found in a subset of our keratoconus population, we have not found a single case where chronic and abnormal eye rubbing was not eventually present in the patient’s history.
The importance of the sleeping habit
As others have observed (59), we have noticed that patients with keratoconus are more susceptible to sleeping on their stomach or on their side. Because of the constrained lateral head position, they typically exert an extended pressure against one or both orbits while they sleep. Some patients favor the right or the left side when they fall asleep. The strong correlation between the side which is more compressed in the pillow/arm/hand at night and the side of the most advanced keratoconus suggests that even the sleeping habit may play a significant role in the genesis of keratoconus. In this context, asymmetrical eye rubbing may be again the confounding variable. The direct and prolonged contact of the eyelids against the linen may increase the contamination of the ocular surface by irritants and allergens such as dust mites, contributing to increased local pruritis and subsequently increased eye rubbing of the involved eye. In my practice, the correlation between keratoconus and such sleeping habits is striking and even superior to the correlation with the dominant hand which has been previously reported only in severe keratoconus (60).
With all of this knowledge in mind, designing a prospective study including patients randomly assigned to rub (or not) their eyes for extended periods of time may be contraindicated for ethical reasons. Hence, these observations (a comprehensive case reports will be soon presented) have a significant evidence-based function in assessing the causal hypothesis of eye rubbing being the root and therefore necessary cause of keratoconus.
Conclusion
In this article, I have presented many bodies of evidence which strongly support the conjecture that chronic eye rubbing and its mechanical influence on corneal tissue appears to be the only necessary inciting event for the permanent corneal warpage clinically designated as “keratoconus”.
Chronic eye rubbing elicits structural changes to the cornea via the forces applied against the corneal stroma, and triggers molecular cascades and cellular activation leading to a reduction of stromal resistance. Keratoconus is not inherited stromal dystrophy and the corneal deformation is not ectatic but isometric and corresponds to permanent corneal warpage induced by excessive eye rubbing.
Accepting this conjecture contradicts the widely accepted idea that this disease is rooted in hidden genetic mechanisms. However, this conjecture provides a better explanatory framework to the genesis and clinical presentation of keratoconus. Without any other particular condition, there is ample evidence that pathological eye rubbing can induce a permanent corneal deformation via various but synergistic mechanisms. Eye rubbing is the least common multiple of many conditions predisposing to keratoconus: while some patients may be predisposed to keratoconus via a constitutively thinner or softer cornea, it is important to realize that only when a repeated mechanical force is applied against the cornea that the latter will undergo permanent focal deformation, leading to the classic presentation of keratoconus.
That every patient presenting with keratoconus has rubbed one (unilateral presentation) or both eyes seems a challenging assertion which is equally difficult to approve or refute. However, the accumulated body of evidence and the compelling clinical investigations speak vehemently to consider eye rubbing as a necessary ingredient in the “keratoconus recipe”.
In our experience, patients who cease to rub their eyes do not show any progression. The cessation of eye rubbing brings about the stabilization of the cornea: beyond the offered clinical benefit, it is another strong argument in favor of the “no rub, no cone” conjecture. As I am not involved in pediatric keratoconus, my keratoconic patients are all of the adult age (16 yo and higher). A recent meta-analysis has shown that pediatric keratoconus is more aggressive than adult keratoconus, with high documented rates of progression in pediatric populations (57). In addition, the literature overwhelmingly shows higher rates of failure and progression of corneal collagen cross-linking, intracorneal ring segments and penetrating keratoplasties as compared to adults. While this could be explained by structural differences in the cornea between the two populations, the reduced compliance and the persistence of irrepressible eye rubbing in children could also account for the aggressiveness of pediatric keratoconus. However, considering that eye rubbing is the indispensable trigger precipitating the cascade of events leading to permanent corneal warpage paves the pathway towards keratoconus prevention in children and young adults.
This primarily mechanical theory can unify most of what is observed and be coherent with the results of recent clinical and experimental investigations, the “no rub, no cone” conjecture should be regarded as an important milestone in our understanding of the keratoconus physiopathology.
References


Firstly, it is important to keep in mind that not every chronic eye rubber will develop keratoconus. It is a matter of intensity x duration x frequency of the rubbing on one side, and of the native corneal resistance on the other. However, patients who rub their eyes often and vigorously exhibit some minor corneal irregularities, which have been coined by some « forme fruste KC », as it was believed that there would be such condition as an abortive or non-evolutive form of KC. Corneal topography is mandatory to confirm the presence of such topographic irregularity. It is also mandatory to stop rubbing your eyes after corneal refractive surgery. Asian eyes are not susceptible to develop more symptoms or complications after refractive surgery, and they are eligible for having any technique, depending on the preoperative evaluation result.
Good evening Dr. Gatinel,
Happy New Year and thanks for allowing me to repost it here, I hope this is a more relevant column .
I am 32y, East-Asian with myopia & astigmatism (-1.75 (-0.75),-1.75 (-1.25)). I am heavy computer screen user (>12h per day), and I haven’t been wearing glasses systematically(<1h per day) until very recently, after having experienced frequent eyestrain and possible eyesight drop.
Initially I went to your website to learn more about the laser surgery then I discovered about your article on the linkage between keratoconus and eye Rubbing.
I realized immediately that I'm guilty of having mentioned bad habit. I was systematically pressing my eyes when I was at 20s because I thought my eyes were "popping out". I also do not wear glasses or contact lenses.
I have a couple of questions :
How can we separate the keratoconus of an early stage from "normal" Astigamstim if topography exam is not systematicly proposed? (I had ophthalmologist appointment in 2018 but topography test was never even mentioned.)
Will high-risk-behavior population (like myself) be considered as sub-clinical KC automatically and by consequence non eligible for lasik ?
Asian eyes differs in shape to Europeans eyes with a thicker cornea. Will this increase the chance of mis-diagnosing KC (false negative. ex) ?
I was told that Asian eyes will experience more severe and long lasting post-surgery symptoms such as dry eyes and pains. is this true? Will the lasik/pkr/smile performed in Europe be suitable for Asian eyes?
Do you have any good colleague/hospital in Belgium that you can recommend for KC screening &lasik?
I wish I had read your article about eye rubbing 20 years earlier, I m very grateful for your effort of sharing this so valuable information. Thank you in advance for your patience and good vibes.
Many thanks
Bonsoir,bonne continuation dans vos recherches et merci!!